|
Some tables are available as slides in Google Apps. Metals are good conductors because they have. The transition elements, on the other hand, lose the ns electrons before they begin to lose the ( n – 1) d electrons, even though the ns electrons are added first, according to the Aufbau principle. This is a collection of free printable periodic tables in PDF file or PNG image format to save, print, and use. The number of valence electrons in an atom is reflected by its position in the periodic table of the elements. As a general rule, when the representative elements form cations, they do so by the loss of the ns or np electrons that were added last in the Aufbau process. Thus, as we would expect, the outermost or valence electrons are easiest to remove because they have the highest energies, are shielded more, and are farthest from the nucleus. Likewise, most atoms with 1, 2, or 3 valence electrons can lose electrons. 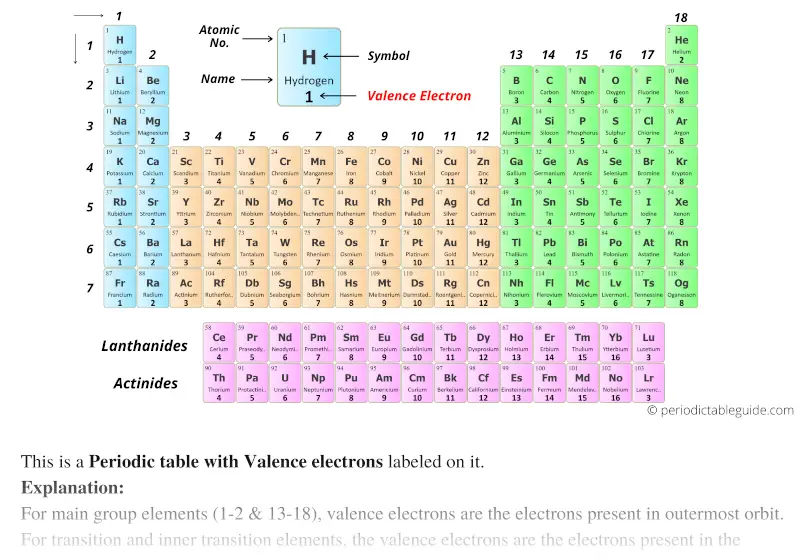
The locations of period 4 and period 5 transition elements are provided with green dots. Similarly, minima exist at the locations of noble or inert gases: H e, N e, A r, K r, X e, and R n. The electrons in the outermost shell are the valence electrons the electrons on an atom that can be gained or lost in a chemical reaction. Peaks are evident at the locations of the alkali metals: L i, N a, K, R b, and C s. Although astatine is radioactive and only has. These five toxic, non-metallic elements make up Group 17 of the periodic table and consist of: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). The halogens are located on the left of the noble gases on the periodic table. periodic table, PS1.A, SEP2, valence, valence electrons. Group 17: General Properties of Halogens. A black jagged line connects the radii values for elements with atomic numbers 1 through 60 on the graph. Lewis Structures Notes & (pdf) Additional review Practice. Here Ne refers to the core electrons which are the same as for the element neon (Ne), the last noble gas before phosphorus in the periodic table. The number of valence electrons in an atom is reflected by its position in the periodic table of the elements (see the periodic table in the Figure below). This graph is labeled Atomic Number on the horizontal axis and Radius (p m) on the vertical axis. Valence Electrons and the Periodic Table. Within each group (e.g., the alkali metals shown in purple), the trend is that atomic radius increases as Z increases. Without using a periodic table or any other references, fill in the correct box in the periodic table with the letter of each question. \): Within each period, the trend in atomic radius decreases as Z increases for example, from K to Kr.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
